Glaucoma is a serious eye disease that can gradually cause vision loss, which is currently irreversible. But in a new cell culture study scientists found that removing a membrane at the back of the eye could help transplanted cells migrate into the optic nerve and repair the connections, potentially restoring lost vision.
Retinal ganglion cells sit at the back of the eye and make up the optic nerve with their long tendril-like axons, transmitting visual information to the brain. But the inner eye pressure associated with glaucoma can damage these cells, and if left untreated even kill them, resulting in vision loss.
“That’s why it is so important to detect glaucoma early,” says Thomas Johnson, an author of the new study. “We know a lot about how to treat glaucoma and help nerve cells survive an injury, but once the cells die off, the damage to someone’s vision becomes permanent.”
For the new study, researchers at Johns Hopkins investigated ways to repair damage to optic nerve cells by growing and implanting new cells. First the team grew mouse retinas in lab dishes, then introduced human retinal ganglion cells to see how well they might integrate.
Unfortunately, most of the transplanted cells failed to integrate with the retinal tissue, clumping together instead of dispersing. But there was one key exception: several small groups managed to integrate properly in certain areas.
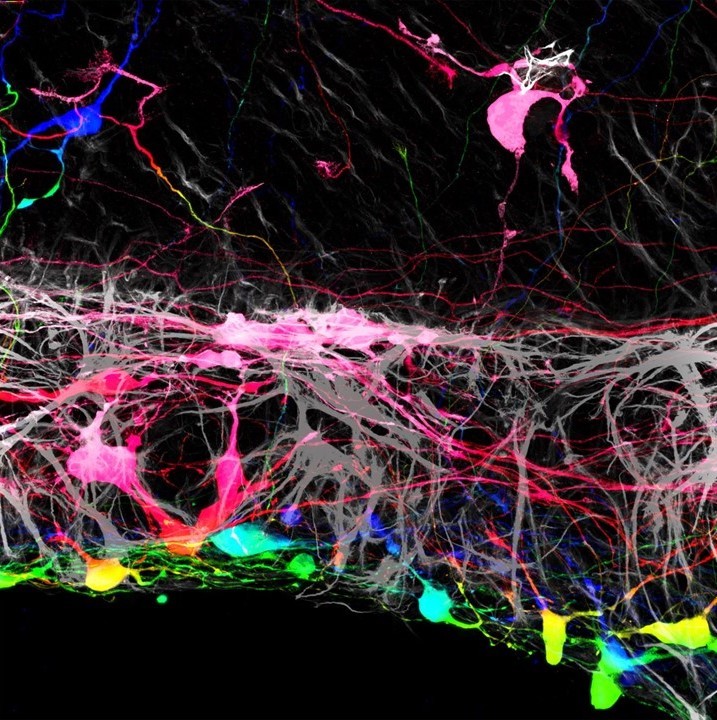
Thomas Johnson and Johns Hopkins Medicine
When the team investigated more closely, they found an intriguing pattern behind why the cells settled in some places and not others. The new cells were slipping through incisions that the researchers had made in the retinas, in order to get them to lie flat in the dish.
“This suggested that there was some type of barrier that had been broken by these incisions,” says Johnson. “If we could find a way to remove it, we may have more success with transplantation.”
The culprit turned out to be the internal limiting membrane (ILM), a layer that separates the retina from the vitreous body – the clear gel-like substances that fills the eyeball. So in follow-up tests, the researchers loosened its connective fibers using enzymes, removed the membrane and then transplanted new retinal ganglion cells. Sure enough, without that barrier in the way, most of the transplanted cells managed to integrate properly, even establishing new nerve connections.
“These findings suggest that altering the internal limiting membrane may be a necessary step in our aim to regrow new cells in damaged retinas,” says Johnson.
Of course, one major question looms over the study: is it safe to remove the internal limiting membrane? Currently ILM “peeling” is a surgical practice used to help improve outcomes in macular hole treatment – although the potential side effects make it a subject of debate. If removing the ILM can restore vision loss previously considered irreversible, then perhaps it’s worth trying. Either way, this is still very early days for the research, and the team plans to continue investigating the technique.
The study was published in the journal Stem Cell Reports.
Source: Johns Hopkins Medicine
Source of Article