Projections from the Centers for Disease Control and Prevention (CDC) estimate the new SARS-CoV-2 variant XBB.1.5 has exploded in prevalence across the United States, rising from just 3% to over 40% in a few short weeks. The variant is thought to have emerged in New York and researchers claim early studies indicate it can more effectively bind to human cells than prior variants.
As the world celebrates the dawn of a new year, and we enter the fourth year of the pandemic, yet another twist in the COVID story has appeared with the emergence of XBB.1.5, a new Omicron subvariant that seems to be rapidly spreading across the United States. The variant looks to be the first recombinant mutation to have any relevant impact on the trajectory of the pandemic.
A recombinant is a virus that is a mix of genomes from different variants. They can emerge when a person is infected with two or more variants simultaneously.
“When a virus copies itself, it can ‘template switch’ between different genomes floating around,” explained researcher Emma Hodcroft. “If those are all the same the end product isn’t different! But if distinct lineages are there at the same time, we can spot these ‘chimera’ children – recombinants!”

Plenty of SARS-CoV-2 recombinants have appeared in recent times, often resulting in hyperbolic headlines heralding the threat of a “deltacron” strain emerging. However, until very recently, no SARS-CoV-2 recombinant has ever spread significantly enough to generate a relevant infection wave.
The first recombinant to make a serious appearance arose a few months ago and was dubbed XBB. The unique variant is a combination of BJ.1 (aka BA.2.10.1.1) and BM.1.1.1 (aka BA.2.75.3.1.1.1).
XBB was initially identified in India in August and Singapore was the first country to experience an XBB wave in October. That wave peaked relatively quickly without causing too much trouble to the country’s healthcare system, suggesting XBB may not cause too much trouble worldwide and leading many researchers to breathe a sign of relief.
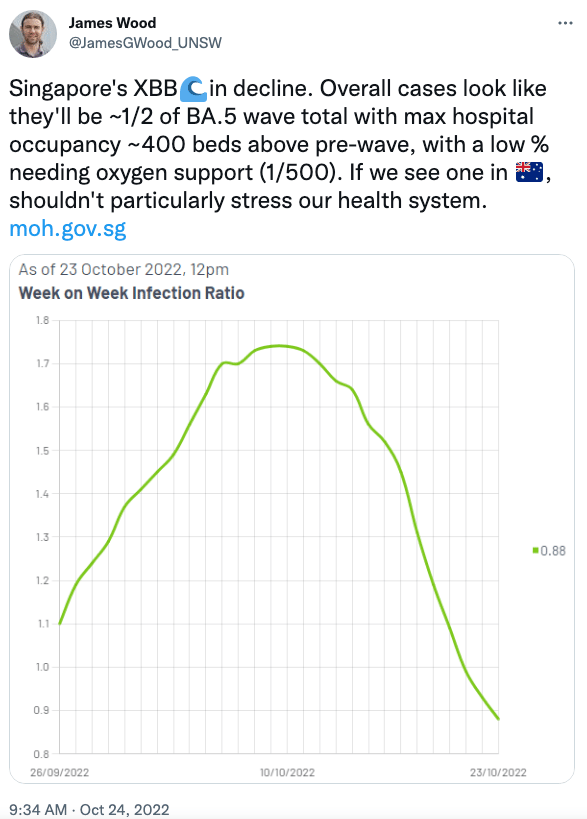
But the last year has demonstrated no SARS-CoV-2 variant stays the same for too long, and as XBB travelled the world it quickly picked up new mutations. Enter XBB.1.5, possibly the first variant of concern to emerge out of the United States.
The first reported XBB.1.5 sample has been traced back to the 22nd of October in New York. A week later the variant was detected in Rhode Island and Washington state. Scattered detections were then traced in India and Indonesia in early November but by the middle of the month a number of European countries were picking up the new variant.
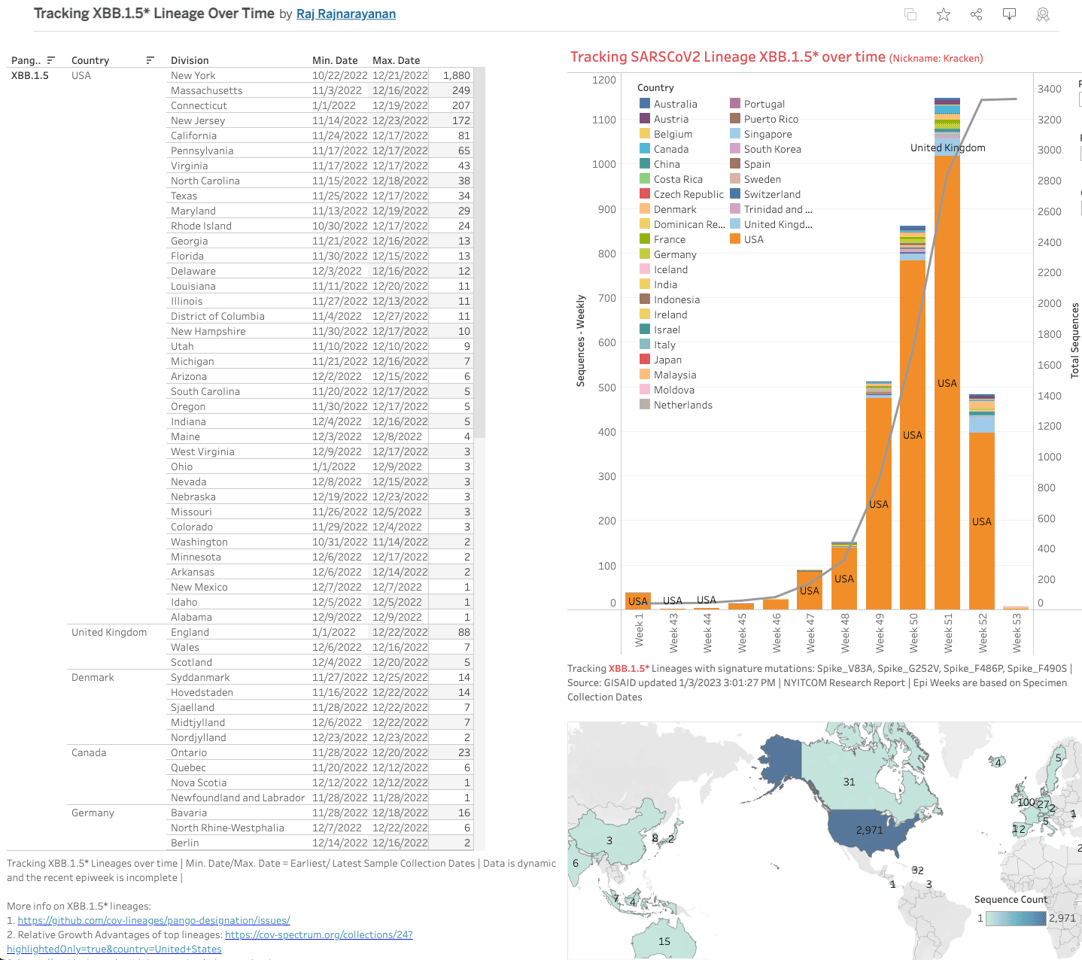
Until last week the CDC’s variant tracking model had not separated XBB.1.5 from XBB. But just before the turn of the new year its new forecast appeared, and suddenly XBB.1.5 had been included, projected as accounting for more than 40% of all cases in the United States.
Due to the slow pace of genomic sampling, the CDC employs a modeling system called Nowcast to predict variant changes in real-time. The current Nowcast model estimates XBB.1.5 cases have doubled in the US over the past week, and in the northeast of the country account for more than 75% of all confirmed cases.
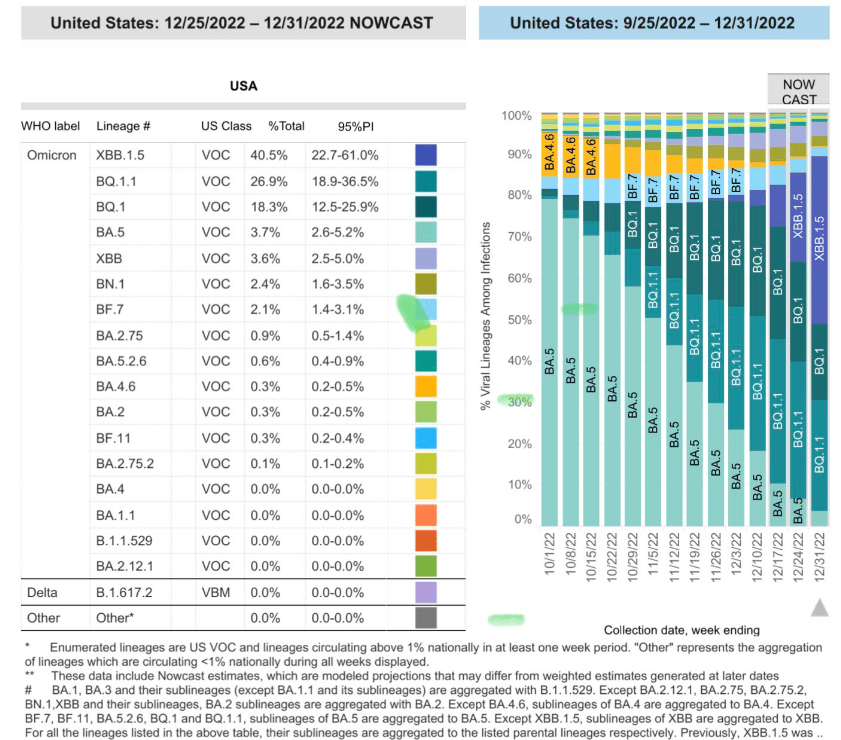
CDC
As always, the million-dollar question is whether XBB.1.5 causes more severe illness than prior Omicron variants. Eric Topol, from the Scripps Research Institute, said New York can be considered a bellwether for XBB.1.5, and the region has seen a concerning rise in hospitalizations over the last few weeks. According to Topol this spike in hospitalizations may not be entirely due to the rise of XBB.1.5, but the new variant is almost certainly playing some kind of role.
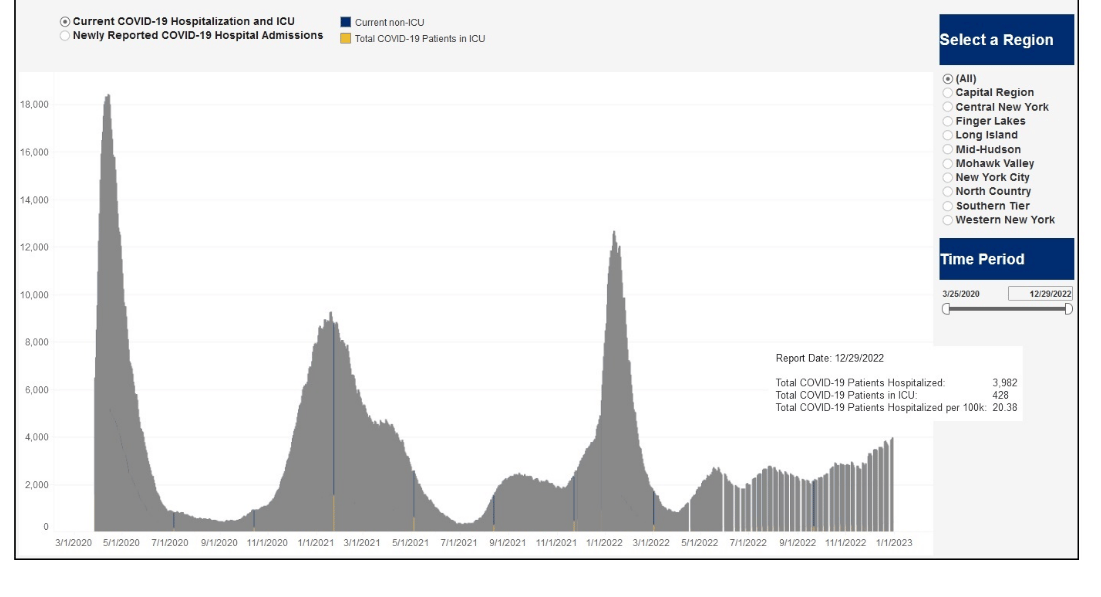
“Of course, other factors are likely contributing such as waning of immunity, indoor/holiday gatherings, cold weather, lack of mitigation,” Topol explained on his blog. “But it is noteworthy that New York’s Covid hospital admission rate is the highest since late January (and also exceeds the summer 2021 Delta wave, but with some ambiguity as to how hospitalization were categorized then and now).”
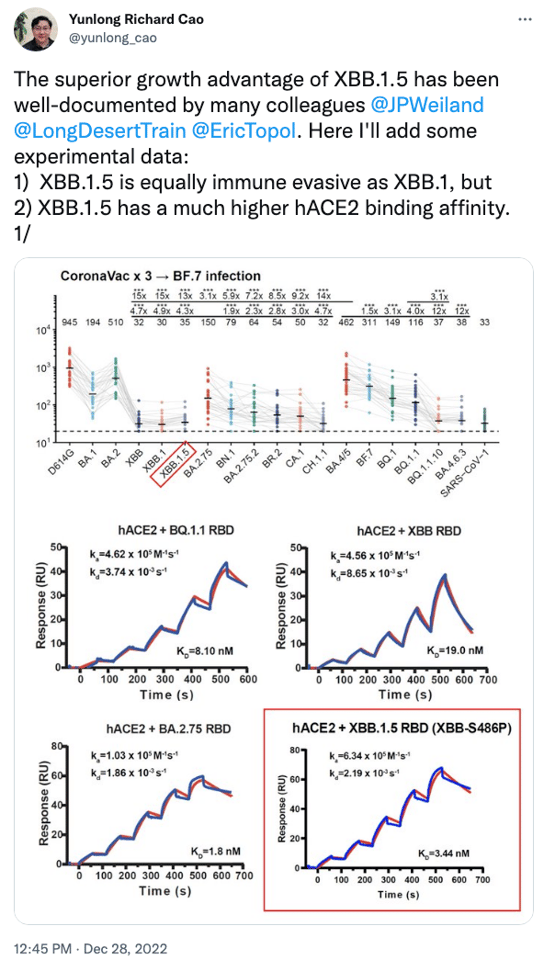
Alongside these epidemiological observations new experimental data from researcher Yunlong Cao indicate a key mutation in XBB.1.5 may explain how this variant can more effectively bind with human cells. Dubbed F486P, the mutation enhances the virus’s ability to bind with certain receptors on human cells. While other mutations help this particular variant evade immune antibodies developed from prior infections, F486P means the virus can more efficiently get into human cells.
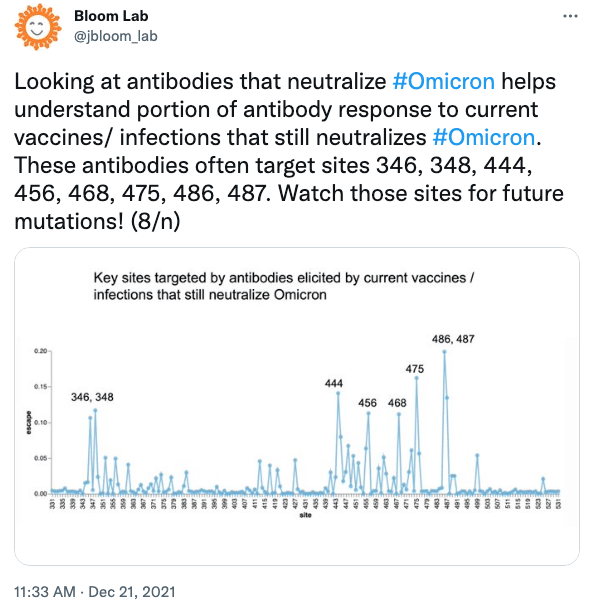
Around a year ago Jesse Bloom, a researcher tracking the evolution of viruses, flagged 486 as a key genomic site for future problematic SARS-CoV-2 mutations. Current vaccine- and infection-induced antibodies specifically rely on that spot to effectively neutralize the virus.
So most mutations at site 486 lead to a more immune evasive variant, but it’s evolution at a cost. Studies linked earlier 486 mutations to less effective human cell receptor binding. This means earlier SARS-CoV-2 variants sacrificed cellular binding for antibody evasiveness.
The big difference between XBB.1.5 from its parent lineages (XBB and XBB.1) is that it has traded a F486S mutation for F486P, and F486P is the only 486 site mutation that straddles the best of both worlds, antibody escape without sacrificing efficient human receptor affinity.
“Therefore, XBB.1.5 isn’t expected to have more antibody escape than XBB.1 (which already had mutated F486), but it should have greater ACE2 affinity,” Bloom recently explained. “So it’s greater ACE2 affinity (and perhaps RBD [receptor binding domain] stability) that is giving XBB.1.5 its boost in transmissibility, and causing it to surge.”
Yunlong Cao also speculates the unique cellular binding affinity of XBB.1.5 could make it more susceptible to generating further novel mutations in the future. So, of course, XBB.1.5 is unlikely to be the end of the road for SARS-CoV-2 variants.
“Another important observation is that XBB.1.5’s hACE2 binding affinity is almost comparable to that of BA.2.75, which may enable XBB.1.5 to gain more mutations, similar to what BA.2.75 had,” Cao suggested on Twitter. “It’s just XBB.1.5 haven’t felt much immune pressure yet.”
None of this means XBB.1.5 will necessarily generate more severe disease than prior variants but it does mean XBB.1.5 may be able to infect more people, more easily, and subsequently track down those most vulnerable to COVID-19 – namely, the elderly, the under-vaccinated and the immunocompromised. XBB.1.5 in no way takes us back to square one but it certainly could prolong the pandemic, spiking infections and again placing pressure on health care systems.
Another potential concern of XBB.1.5 is its impact on long COVID. Yale researcher Akiko Iwasaki noted the increased cellular binding affinity for this new variant could hypothetically lead to a new wave of long COVID infections due to the virus’s enhanced ability at infecting human cells.
“I’m concerned because of the putative ability of XBB.1.5 to have increased capacity to infect cell types that express even lower levels of ACE2,” Iwasaki tweeted recently. “This will increase tropism and possibly persistence in cell types that are long lived.”
Vaccine scientist Peter Hotez said XBB.1.5 raises plenty of new questions that need to be answered but we shouldn’t be fearful or overreact. He points to a recently published study showing the new bivalent COVID vaccine is still effective against newer variants such as XBB. So XBB1.5 may have increased cellular binding affinity but our vaccines are still likely to be useful at reducing disease severity.
Should we be looking to add a new target to update the vaccine in 2023, Hotez asks. That’s a question that scientists and variant trackers will invariably be asking as the new year progresses and SARS-CoV-2 continues to shape-shift around our defenses.
“I was hopeful 2022 would work out better than 2021,” Hotez wrote on Twitter. “Maybe a little, but not so much… now hoping 2023 works out better than 2022 did.”
1/n Regarding XBB 1.5 seeing lots of things online but I’m reminded of Dr Curie’s quote. We need to better understand the following… https://t.co/Jk2GPhCcp6
— Prof Peter Hotez MD PhD (@PeterHotez) December 31, 2022
Source of Article