As important as the CRISPR-Cas9 gene-editing tool has been, it has a relatively high error rate, which can introduce potentially harmful mutations. Researchers in Germany have now developed a more refined tool that reduces errors by nicking DNA instead of cutting it.
CRISPR is one of the most groundbreaking inventions of the century, with the potential to revolutionize gene therapy for a range of diseases, as well as improving the yield or nutrition of crops, creating beneficial microbes, and a variety of other uses. It works like a pair of “molecular scissors,” cutting out problematic genes and slotting in more helpful ones.
The problem is, sometimes it can target the wrong section of DNA and make a change there – known as an off-target mutation – which can potentially trigger health issues. Even if it does get the right target, the DNA repair process can go wrong and lead to what’s called an on-target mutation.
Preventing both of these issues was the focus of the new study, by researchers at the Max Delbrück Center for Molecular Medicine (MDC) and Humboldt University of Berlin. The team modified these molecular scissors to be a bit more gentle, making a fundamentally different type of cut.
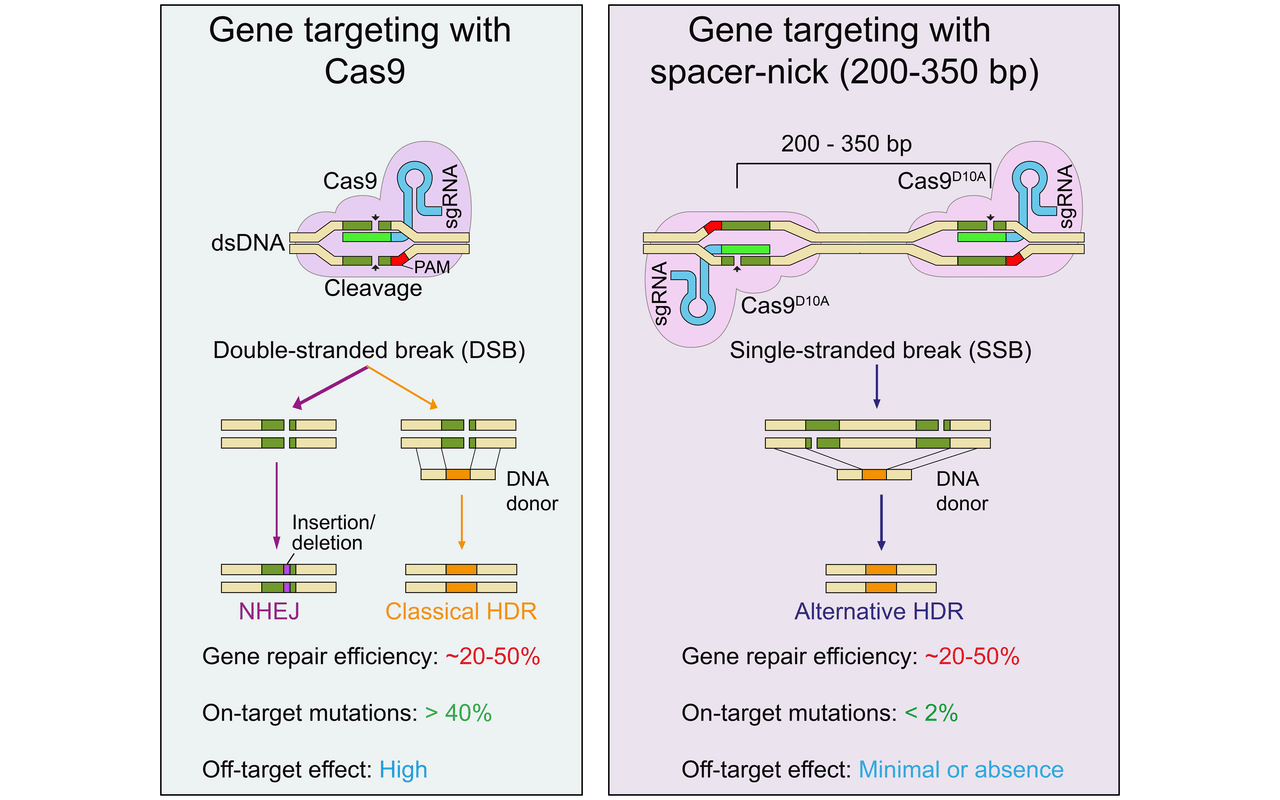
Rather than making one cut that slices right through the entire double strand of DNA, the new tool makes two smaller nicks, each cutting one DNA strand. A built-in spacer keeps those nicks a safe distance apart – between 200 and 350 base pairs.
“Our experiments with hematopoietic stem cells and T cells have shown that this is the optimal distance for minimizing both on-target and off-target mutations,” said Dr. Van Trung Chu, co-last author of the study. “Any shorter, and we risk cutting through the entire DNA molecule – despite the use of two separate scissors.”
Van Trung Chu, MDC
In tests on cells in lab dishes, the team found that the new spacer-nick tool was about as effective at making edits as conventional CRISPR – between 20 and 50 percent of treated cells were repaired. But importantly, the new tool was much better at reducing errors, with on-target mutations occurring in under two percent of edits with spacer-nick, compared to over 40 percent for CRISPR-Cas9. Off-target mutations, meanwhile, appeared to be “a rare, if not non-existent, occurrence in our approach,” said Chu.
The team hopes that future work will test the spacer-nick editing tool in animals, before moving onto human trials. One of the first potential targets is to treat inherited blood disorders.
The research was published in the journal Science Advances.
Source: MDC
Source of Article